For the first time since 2019, ~31,000 oncologists attended the 2022 ASCO Annual Meeting in person, recapturing the energy of the pre-Covid era. While new data that can transform patient care in oncology continued to emerge, cell therapies were among the early adopters. Cell therapy players had a successful ASCO 2022, with many developments to address challenges and generate efficacy in high unmet needs and low immunoresponsive tumors. Key presentations to watch include IN8 Bio’s gamma delta T cells, Instill Bio’s adoptive TILs, Innovative Cellular Therapeutics’ coupled CARs, and data from NY-ESO-1 targeted TCR T cell therapies. It is important to note that cell treatment with lovance’s lifileucel has progressed to the most advanced stage in solid tumors. The drug is currently undergoing BLA filing for advanced melanoma. The company plans a pre-BLA meeting in July 2022 to submit the application by August 2022.
Report #2
Cell therapies in solid tumors: Early steps to measured strides in low immuno-responsive tumors
Our report #1 highlighted ASCO 2022 data of cell therapies in heme malignancies – New targets & innovations do not let the CAR lights dim in Heme
In contrast to heme, the development of cell therapies in solid tumors is still in nascency due to a variety of reasons, including tumor heterogeneity, paucity of tumor-specific antigens, immunosuppressive tumor microenvironment (TME), and potentially fatal on-target, off-tumor toxicities. However, researchers are coming up with renewed strategies to address these issues. Continuing the trends of the last few years, at ASCO 2022 also, pharmaceutical companies and academia were spotted investigating novel cell sources and approaches to resolve the issues cell therapies encounter in solid tumors. Besides CAR-Ts, studies demonstrated the potential of gamma delta T cells, TCR T cell therapy, and adoptive TILs in high unmet need and low immuno-responsive tumors where established checkpoint inhibitor therapies have failed to generate optimal responses e.g. soft tissue sarcoma, GBM, CRC, and PD-1 refractory melanoma.
Adoptive TILs are the most advanced cell therapy programs: continued promise in checkpoint inhibitor refractory melanoma
 Adoptive therapy with TILs (Tumor Infiltrating Lymphocytes) has shown the ability to generate complete responses in patients with solid tumors who have failed to respond to standard therapies. Impending BLA for Iovance’s lifileucel in metastatic melanoma and recent encouraging early data of Instil Bio’s adoptive TILs, ITIL-168, not only validate their potential as a valuable therapeutic for patients but also as a platform for innovation and next-generation therapies. A single-center use clinical trial demonstrated the feasibility and clinical utility of an earlier version of ITIL-168, with a high ORR (58%, N = 12) in patients previously treated with PD-1 inhibitor.
Adoptive therapy with TILs (Tumor Infiltrating Lymphocytes) has shown the ability to generate complete responses in patients with solid tumors who have failed to respond to standard therapies. Impending BLA for Iovance’s lifileucel in metastatic melanoma and recent encouraging early data of Instil Bio’s adoptive TILs, ITIL-168, not only validate their potential as a valuable therapeutic for patients but also as a platform for innovation and next-generation therapies. A single-center use clinical trial demonstrated the feasibility and clinical utility of an earlier version of ITIL-168, with a high ORR (58%, N = 12) in patients previously treated with PD-1 inhibitor.
Based on promising results, phase II DELTA-1 trial has been initiated to evaluate the efficacy and safety of ITIL-168 in melanoma patients relapsed or refractory to a PD-1i, pts intolerant to a PD-1i, and those who had stable disease as the best response to a PD-1i.
Source: Abs#TPS9594, Poster# 185b
Cell therapies demonstrate efficacy as salvage treatment across gastro-intestinal tumors
 CARsgen Therapeutics’ Claudin 18.2 CAR-T appears promising in gastric cancer
CARsgen Therapeutics’ Claudin 18.2 CAR-T appears promising in gastric cancer
CARsgen Therapeutics presented two posters for their Claudin 18.2, targeted autologous CAR T-cell therapy (CT041). Claudin18.2 is a validated target overexpressed in gastric and pancreatic cancers. The 1st poster (abs # 2538) presented results from the multicenter phase 1b CT041 trial in the U.S. for patients with advanced gastric and pancreatic adenocarcinoma, while the 2nd poster (abs #4017) presented safety and preliminary efficacy results from the Phase Ib/II CT041 study in China for patients with advanced gastric/gastroesophageal junction adenocarcinoma. The US trial (phase 1b trial (NCT04404595)) reported an ORR of 60% (N=5) with 1CR in heavily pre-treated (≥ 2 prior lines) in GC/GEJ patients. ORR compares favorably against current therapies in the setting. No responses, however, were observed in the pancreatic cancer cohort (N=7).
The China Study (phase Ib/II, NCT04581473) in patients with advanced GC/GEJ adenocarcinoma who were refractory to or intolerant of at least 2 prior treatments also reported promising outcomes with 57% ORR. At a median follow-up of 8.8 months, the mPFS and mOS were 5.6 months and 10.8 months respectively. CT041 was well tolerated in both studies. No DLTs, ICANS, or Tx-related deaths were observed. Most grade1- 2 CRS was noted except for one case of grade 4 CRS in the China study.
Source: Abstract #4017, Poster Bd #5, Abs#2538
Coupled CAR’s two-pronged approach to tumor-killing wins in CRC
Innovative Cellular Therapeutics Inc. presented a unique approach of coupled CARs in which one of the CAR- T mediates cell proliferation while the other causes tumor killing.  The company provided an update of the lead product from this CoupledCAR® technology, guanylate cyclase C (GUCY2C) targeted, GCC19 CART being developed for relapsed/refractory CRC. Coupled CAR combines solid tumor CAR-Ts with CD-19 CARTs. The technology relies on the fact that the killing of B cells by CD19 CAR-T cells triggers the immune system and results in the proliferation of all T cells, including any non-transduced T cells and solid tumor CAR-T cells without the necessity for the solid tumor CAR-T cells to bind to the target antigen. The release of cytokines by Coupled CAR promotes solid tumor CAR-T cell infiltration and killing of target cells. In a phase 1 trial, 21 patients have been dosed at two dose levels (1×106 cells/kg, N=13 and 2×106 cells/kg, N=8 ). Overall ORR was 29%, higher responses were seen at dose level 2 (2×106 cells/kg) where ORR was 50%. Median survival at both the doses was >10 months and compares favorably against historical records with the SoC (regorafenib or Lonsurf- mOS 6.4-7 mos). The most common adverse events were cytokine release syndrome (CRS) in 20/21 subjects, though grade 3 was observed in only one patient. ≥Grade 3 neurotoxicity was noted in two cases and resolved with corticosteroids.
The company provided an update of the lead product from this CoupledCAR® technology, guanylate cyclase C (GUCY2C) targeted, GCC19 CART being developed for relapsed/refractory CRC. Coupled CAR combines solid tumor CAR-Ts with CD-19 CARTs. The technology relies on the fact that the killing of B cells by CD19 CAR-T cells triggers the immune system and results in the proliferation of all T cells, including any non-transduced T cells and solid tumor CAR-T cells without the necessity for the solid tumor CAR-T cells to bind to the target antigen. The release of cytokines by Coupled CAR promotes solid tumor CAR-T cell infiltration and killing of target cells. In a phase 1 trial, 21 patients have been dosed at two dose levels (1×106 cells/kg, N=13 and 2×106 cells/kg, N=8 ). Overall ORR was 29%, higher responses were seen at dose level 2 (2×106 cells/kg) where ORR was 50%. Median survival at both the doses was >10 months and compares favorably against historical records with the SoC (regorafenib or Lonsurf- mOS 6.4-7 mos). The most common adverse events were cytokine release syndrome (CRS) in 20/21 subjects, though grade 3 was observed in only one patient. ≥Grade 3 neurotoxicity was noted in two cases and resolved with corticosteroids.
Relapsed /refractory CRC is a high unmet need area with a median survival of just 6-7 months. Immunotherapies such as anti-PD-1/L1 have been generally ineffective except MSI-high segment. The early promise of cell therapies has been observed with CARTs against CEA, HER2, NKG2D, EGFR targets, however, none of them have reached to advanced phase. GUCY2C is a membrane-bound cyclase whose cell surface expression is restricted to the apical surfaces of intestinal epithelial cells and a subset of hypothalamic neurons. Its expression is observed in >95% of CRC metastases and it is ectopically expressed in tumors that evolve from intestinal metaplasia. The restricted expression is expected to provide a safety advantage which has been observed in this trial with only one case of grade 3 CRS and 2 cases of grade 3 neurotoxicity. Mature outcomes in the future will inform the potential of this CAR-T in CRC.
Source: Abstract #3582, Poster Bd #376
Combining NK cell therapy with immune stimulatory therapies betters the SoC in heavily pretreated pancreatic cancer
With three or more prior lines of therapy, the median survival time for patients with pancreatic cancer is only three to four months. Most of the trials conducted in the past have not succeeded in significantly altering this situation. According to immunity bio experts, who presented results from their phase II Quilt trial at ASCO 2022 effective response against pancreatic cancer requires a coordinated approach that orchestrates both the innate and adaptive immune system. They also proposed that immunogenic cell death with lasting responses may be achieved by inducing DAMPs to reveal tumor-related antigens and coordinating the activation of the entire immune system. According to the trial’s findings, The NANT Cancer Vaccine (a combination of DAMP inducers, NK and T cell activation by IL-15 cytokine fusion protein, and PDL1-targeted high-affinity NK cell therapy) nearly doubled survival with mOS of 5.8 months and median PFS of 2.3 months in the population, N=78 (3rd line, 4th line, and 5th line). In the third line specifically (N = 34), the median OS was 6.3 months. Grade 3/4 AEs included Anemia (46%), neutropenia (23%), thrombocytopenia (12%), and were mostly linked to chemotherapy. There were no deaths reported.
Source: Abstract #4147, Poster Bd #132
New CAR-T approaches for Ovarian & Testicular Cancer
Ovarian cancer is hard to treat cancer that, so far, has not responded well to immunotherapies. Recently CAR-Ts have shown promising results in this indication. BioNtech’s BNT211 recently presented encouraging Phase I/II trial results at AACR 2022. Data from 16 patients who received dose levels 1 (1×107 CAR-T cells) and 2 (1×108 CAR-T cells) of CLDN6 CAR-T cells alone or in combination with CARVac vaccine was presented. Patients with Testicular cancer (n=8), ovarian cancer (n=4), endometrial cancer, fallopian tube cancer, sarcoma, and stomach cancer (1 patient each) were included. Of the 14 evaluated patients, six demonstrated a partial response (42%) and five had stable disease with shrinking target lesions (35%). Patients with ovarian cancer (n=2) and testicular cancer (n=4) both experienced responses.
Similarly, researchers from China’s Medical Centre of Shanghai Cell Therapy Group have developed a CAR T cell secreting PD-1 nanoantibodies. The nanobodies target mesothelin. Early data from a phase I trial (N=7 evaluable) was presented. The DCR was observed to be 100%. PFS for two patients with SD were reported to be 5.3 months and 4.1 months. The most common adverse reactions were pruritus, tiredness, nausea, and fever. Following infusion, a significant rise in the number of CAR-T cells in the peripheral blood was observed from days 4 through 14, and an increase in PD-1 nanoantibody levels, INF-IL-6, IL-2, and MCP-1 content was observed from days 1-4.
Source: Abstract #e17535, BioNtech Press Release, Apr 2022
Beyond CAR-T and NK cells, the promise of other cell therapies in aggressive underserved tumors
Gamma-delta T cell companies get a boost as IN8bio’s INB 200 reports efficacy in GBM subtypes
 Adicet’s data presented at ASCO demonstrated the potential of gamma delta cells in heme malignancies. IN8bio is another clinical-stage biopharmaceutical company focused on gamma-delta T cells. The company’s DeltEx platform employs allogeneic, autologous, iPSC, and genetically modified cells to develop therapies for solid and heme tumors. The company is focusing on proprietary gamma-delta T cells engineered for chemotherapy resistance. INB-200 exhibited preliminary but encouraging signals of extending PFS and OS benefit in a phase I trial in patients with newly diagnosed GBM. Amongst GBM subtypes, unmethylated GBM has a relatively worse prognosis than other subtypes. INB 200 was effective in both methylated unmethylated subtypes. To date, six patients have been dosed, and all of them performed better than their projected PFS and OS. None of the patients had any serious adverse events or dose-limiting toxicities.
Adicet’s data presented at ASCO demonstrated the potential of gamma delta cells in heme malignancies. IN8bio is another clinical-stage biopharmaceutical company focused on gamma-delta T cells. The company’s DeltEx platform employs allogeneic, autologous, iPSC, and genetically modified cells to develop therapies for solid and heme tumors. The company is focusing on proprietary gamma-delta T cells engineered for chemotherapy resistance. INB-200 exhibited preliminary but encouraging signals of extending PFS and OS benefit in a phase I trial in patients with newly diagnosed GBM. Amongst GBM subtypes, unmethylated GBM has a relatively worse prognosis than other subtypes. INB 200 was effective in both methylated unmethylated subtypes. To date, six patients have been dosed, and all of them performed better than their projected PFS and OS. None of the patients had any serious adverse events or dose-limiting toxicities.
Source: Abstract #2044, Poster Bd #382
NY-ESO-1 TCR T cell therapies gain ground in soft tissue sarcoma(STS) subtypes; safety profile in different studies may be the deal-breaker
 Cancer Testis antigens are ideal targets for cell therapies given their cancer-restricted expression pattern. NY-ESO-1 is a cancer/testis antigen that is expressed in a variety of tumor types and has demonstrated promise as a cell therapy target. High expression of NY-ESO-1 is observed in synovial sarcoma (generally >70% expression) and myxoid/round cell liposarcoma (MRCLS) (80‒90% expression). Data of two key studies presented at ASCO 22 demonstrated the promise of NY-ESO-1 TCR T cell therapy in STS subtypes.
Cancer Testis antigens are ideal targets for cell therapies given their cancer-restricted expression pattern. NY-ESO-1 is a cancer/testis antigen that is expressed in a variety of tumor types and has demonstrated promise as a cell therapy target. High expression of NY-ESO-1 is observed in synovial sarcoma (generally >70% expression) and myxoid/round cell liposarcoma (MRCLS) (80‒90% expression). Data of two key studies presented at ASCO 22 demonstrated the promise of NY-ESO-1 TCR T cell therapy in STS subtypes.
In one study, affinity-enhanced autologous TCR T-cells (TAEST16001) generated encouraging responses in twelve HLA-A*02:01 soft tissue sarcoma (primarily synovial sarcoma) patients with an ORR of 42%, mDoR of 14.1 and mPFS of 7.2 months in phase I clinical trial. TAEST16001 was well-tolerated with no dose-limiting toxicities; Grade 2 CRS was observed in two cases; no cases of neurotoxicity or other serious AEs were reported. 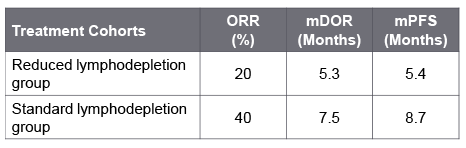 In another pilot study, a single infusion of autologous TCR therapy. Letetresgene autoleucel (Adaptimmune/GlaxoSmithKline) demonstrated antitumor activity among patients with advanced or metastatic myxoid/round cell liposarcoma (MRCLS). The primary analysis showed superior efficacy outcomes among patients who received standard lymphodepletion before treatment with Letetresgene autoleucel compared to a reduced dose. serious TEAEs were observed in nearly half of the patients. 90% of pts had Gr ≥3 TE neutropenia; All grade and grade 3 CRS occurred in 80% and 25% of patients respectively, however, no case of GvHD, ICANs, or Guillain-Barré Syndrome was reported.
In another pilot study, a single infusion of autologous TCR therapy. Letetresgene autoleucel (Adaptimmune/GlaxoSmithKline) demonstrated antitumor activity among patients with advanced or metastatic myxoid/round cell liposarcoma (MRCLS). The primary analysis showed superior efficacy outcomes among patients who received standard lymphodepletion before treatment with Letetresgene autoleucel compared to a reduced dose. serious TEAEs were observed in nearly half of the patients. 90% of pts had Gr ≥3 TE neutropenia; All grade and grade 3 CRS occurred in 80% and 25% of patients respectively, however, no case of GvHD, ICANs, or Guillain-Barré Syndrome was reported.
The investigator of the study Sandra P. D’Angelo, MD, medical oncologist at Memorial Sloan Kettering Cancer Center said that study was guided by prior experience of NY-ESO-1 TCR T cell therapy in synovial sarcoma where promising response rate of 50%, and mOS of ~2 years was observed with the therapy.
Source: Abstract # 11502, Abstract #TPS2681Poster Bd #329b
Engineered macrophages are new addition to cell therapies, however, their clinical value is yet to be established
 Source: Carisma therapeutics
Source: Carisma therapeutics
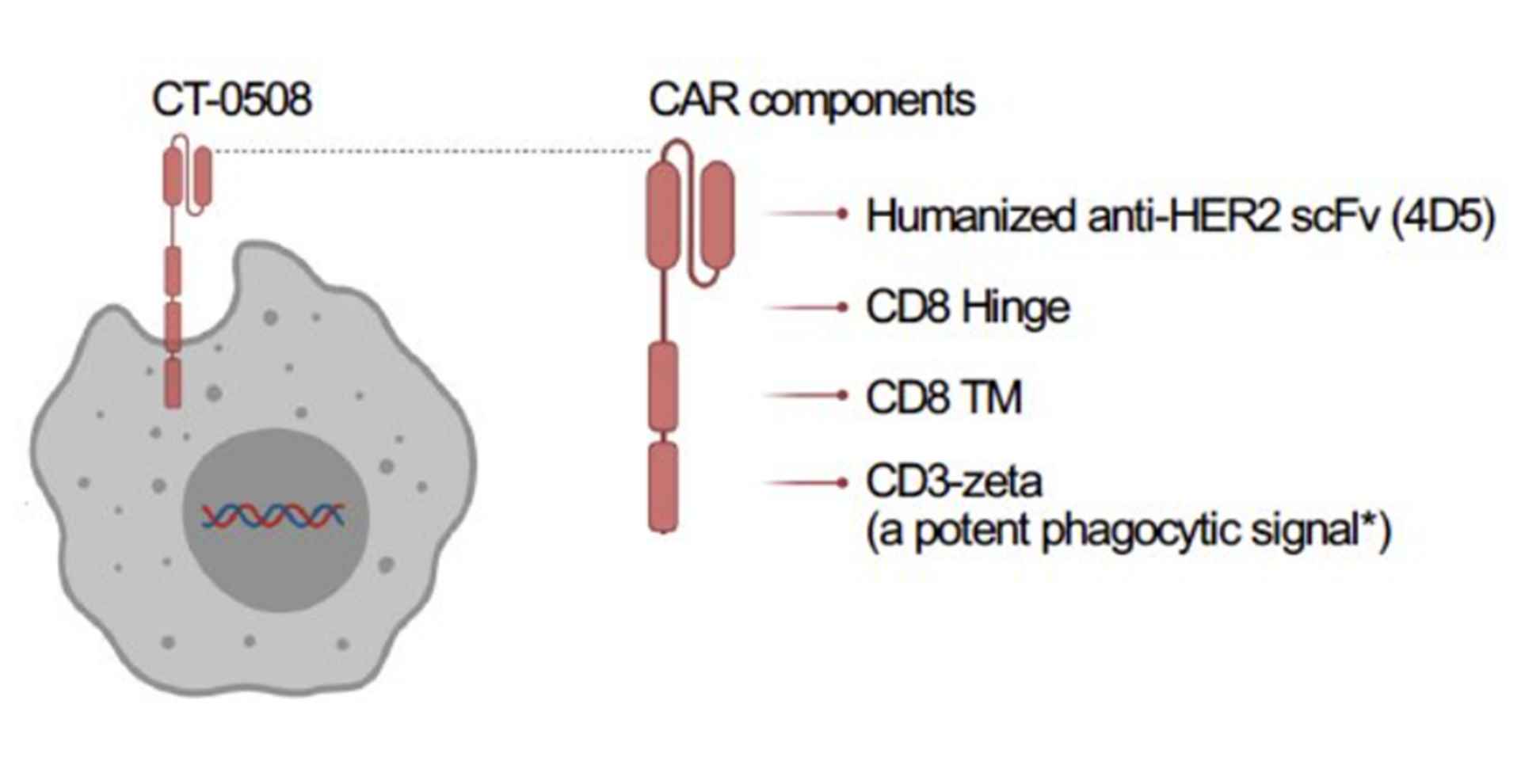
In a very novel approach, Carisma Therapeutics presented the latest data of engineered macrophage therapy CT-0508, from its landmark phase 1 chimeric antigen receptor macrophage (CAR-M) trial in patients with advanced metastatic HER2 overexpressing solid tumors (breast, bladder, pancreatic, ovarian, and stomach cancers). CT-0508 is 1st in class CAR-M comprising of autologous monocyte-derived M1 polarized macrophages expressing anti-HER2. Preclinical studies have shown that CAR-M can infiltrate the tumor, phagocytose the tumor cells, activate the tumor microenvironment, and present the tumor antigens to T cells for their activation and robust anti-tumor responses. CT-0508 was successfully manufactured using macrophages obtained from heavily pretreated, advanced solid tumor patients and showed high CAR expression, viability, and purity. CT-0508 was found to be well tolerated. There were no DLTs, ICANS, or organ toxicities and primarily low-grade CRS. CT0508 exhibited tumor infiltration, remodeling of the tumor microenvironment, and early signs of T-cell infiltration and activation, however, no objective responses were noted. Stable disease was observed as a best overall response.
CT-0508 is 1st in class CAR-M comprising of autologous monocyte-derived M1 polarized macrophages expressing anti-HER2. Preclinical studies have shown that CAR-M can infiltrate the tumor, phagocytose the tumor cells, activate the tumor microenvironment, and present the tumor antigens to T cells for their activation and robust anti-tumor responses. CT-0508 was successfully manufactured using macrophages obtained from heavily pretreated, advanced solid tumor patients and showed high CAR expression, viability, and purity. CT-0508 was found to be well tolerated. There were no DLTs, ICANS, or organ toxicities and primarily low-grade CRS. CT0508 exhibited tumor infiltration, remodeling of the tumor microenvironment, and early signs of T-cell infiltration and activation, however, no objective responses were noted. Stable disease was observed as a best overall response.
Source: Abstract #TPS668, Poster Bd #M10
What is next on the horizon: Safer cell therapies against new targets and neo-antigens
In addition to the above-cited encouraging outcomes, a few biopharma with innovative cell therapies including altered NK cells and T cells that target novel Kita-Kyushu lung cancer antigen-1 (KK-LC-1) cancer/testis antigen presented preliminary findings and trial plans.
NKGen biotech presented data of SNK01 (Autologous Non-Genetically Modified Natural Killer Cells with Enhanced Cytotoxicity) monotherapy in 10 patients from a phase I study in advanced solid tumors. SNK01 showed SD of 60%. There were no major safety issues, and only grade 1 or 2 AEs (abs#2644).
NCI, on the other hand, proposed a phase I trial design (NCT05035407) for the TCR therapy targeting KK-LC-1 (a germ line antigen expressed only in germ cells and overexpressed in gastric cancer, TNBC, cervical cancer, and NSCLC). A cell dosage of 1X108 to 6X1010 TCR-T cells will be given to patients (abs#2678).
Neoantigens are not present in the normal tissues, therefore, they are attractive targets for adoptive T cell therapy. Alaunos Therapeutics presented its neoantigen TCR therapy trial design. An FIH phase I/II of autologous T cells engineered using the Sleeping Beauty transposon/transposase system to express TCRs that are reactive against cancer-specific mutations including those targeting shared KRAS, TP53, and EGFR in patients with advanced solid tumors. Participants in the study are those for whom a TCR that matches the patient’s neoantigen/HLA pairing is available in the TCR library (abs#TPS2679).
Glypican-3 (GPC3) is an antigen that is expressed by more than 75% of HCCs but not by healthy tissues. According to an academic preclinical study presented at ASCO 2022, Natural killer T cells prevent HCC tumorogenesis in mice by interacting with the gut microbiome, and the presence of tumor-infiltrating NKTs is associated with improved clinical outcomes. The findings demonstrated that BATF3 overexpression in NKTs caused significant variations in gene expression profiles when compared to a GFP control both at the start of the experiment and following the destruction of the tumor cells, enriching gene sets involved in metabolic fitness. The results of this study open the path for a phase I clinical trial (Abstract #2644, Poster Bd #298 Abs#TPS2678, Poster Bd #328a)
New innovations and next-generation CAR-Ts, gamma delta T-cells, and highly specific TILs are specifically geared toward solving the issues faced in solid tumors. There is a high probability of cell therapy approval in solid tumors in the near future given heightened competition intensity, strong interest, and positive early findings.
Key questions that will shape the future:
- Out of the diversity of cell sources, which cell therapy class can provide strong benefits with a clean toxicity profile?
- Which cell therapy could be safely positioned in earlier lines?
- What are the novel antigens including intracellular targets and new cancer-testis antigens that can be safely targeted with cell therapies?
- What are the indications where neoantigen-targeted cell therapies have strong potential?
- Which innovations can address multiple challenges in solid tumors?
- Which combination strategy can improve cell therapy outcomes?
- What treatment options can be effective after a CAR-T relapse?
- What are the improvements in manufacturing platforms to make the entire process of cell therapy development more efficient and effective?
FutureBridge Oncology Consulting Services
Our customized oncology consulting services integrate in-depth industry knowledge, relevant-analytics platforms, and PoVs from leading external experts to offer efficient solutions to pharmaceutical and biotech companies in the domains of therapeutics and diagnostics.
Connect with Prakash Dogra (prakash.dogra@futurebridge.com) for any queries or share your business objective here for your project needs.
Need a thought partner?
Share your focus area or question to engage with our Analysts through the Business Objectives service.
Submit My Business ObjectiveOur Clients
Our long-standing clients include some of the worlds leading brands and forward-thinking corporations.
- © 2021 Cheers Interactive (India) Private Limited. All rights reserved. FutureBridge ® is a registered trademark of Cheers Interactive (India) Private Limited.




































