The bone and joint degenerative and inflammatory problems affect millions of people worldwide. They account for half of all chronic diseases in people over 50 years of age in developed countries. Also, it was predicted that the percentage of the population over 50 years affected by the bone disease will double by 2020. Surgery is the common treatment option in such cases. Primary hip and knee arthroplasties belong to the most successful surgeries of this century (>1,000,000/year in the U.S.) and the number of surgeries is rising due to demographical changes¹. These surgeries have a significant positive effect on the quality of life by reducing symptoms and regaining physical function. The number of arthroplasty surgeries is thus increasing every year. In 2010 the 700K (approx.) interventions of total hip arthroplasties were performed. However, this has also led to the rise of revision surgeries and associated complications. Out of all the complications, prosthetic joint infections (PJIs), a type of serious complication of hip and knee arthroplasty, and usually a common cause of revision arthroplasty, are the most feared complications for the patients, as the majority of the PJIs can occur as early as at the time of surgery. It requires a revision of the artificial joint with serious complications which is a cost-intensive procedure. According to a current study, the risk of re-infection after PJI-induced revision surgery is around 8% for hips and knees. Apart from PJIs, fracture-related infections associated with osteosynthetic stabilization are also a major problem as surgeries are often contaminated due to bacterial access by open wounds or fractures. Thus, chronic implant-related bone infections are a serious burden on the health care system.
Medical implants are synthetic devices or prosthetics intended to replace the missing biological structures, support damaged biological structure, or enhance an existing biological structure. These implants are either made of skin, bone, and other body tissues or from metal, plastic, ceramic, or other materials. Implants can be placed permanently or can be removed once they are no longer needed.
Orthopedic implant infection management is crucial as implants can lead to severe infections in the host body. These infections are the result of bacteria adhesion to an implant surface and subsequent biofilm formation at the implantation site. Thus, the treatment of infection is associated with fracture and prevention of chronic osteomyelitis.
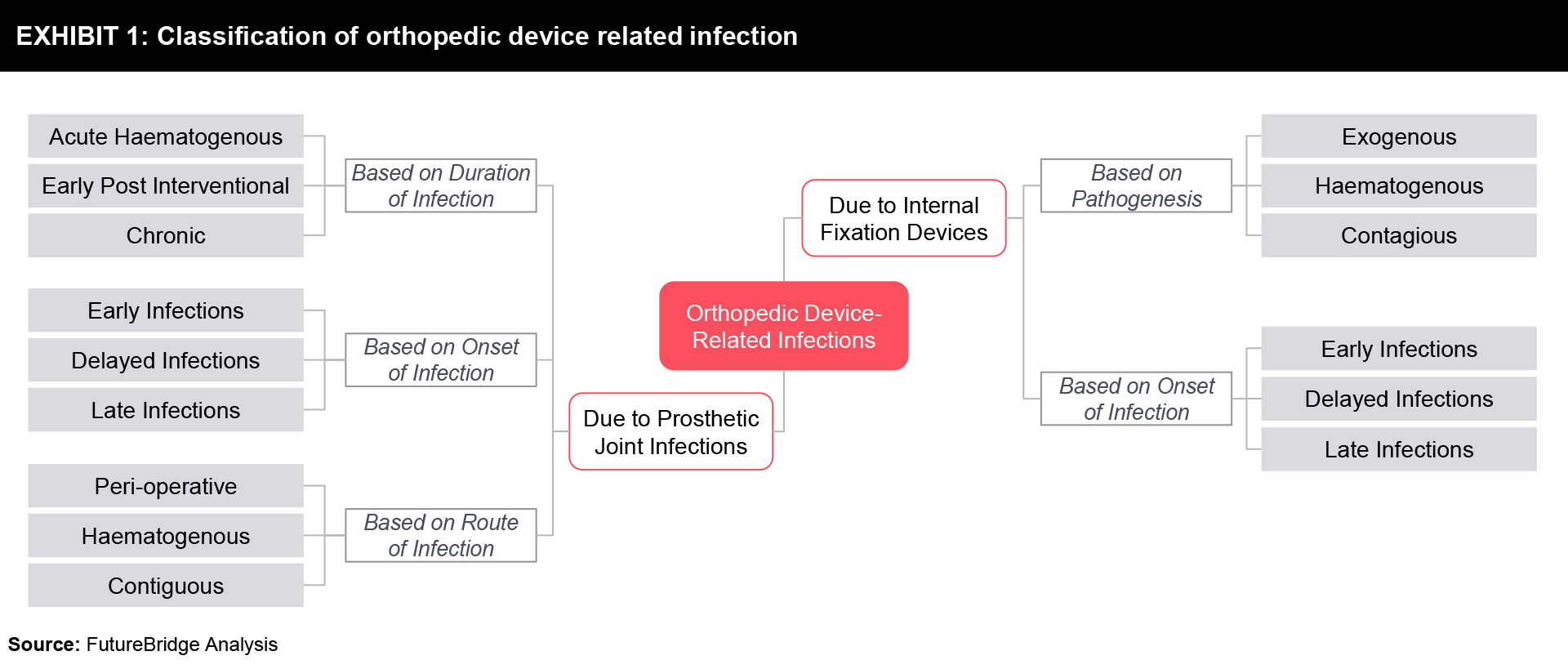
Infections due to Implants
An implant must possess both structural and surface compatibility with the host tissue. Thus, with reference to bone implants, mechanical and physio-chemical compatibility is also required. The orthopedic implant devices are intended to restore the function of loadbearing joints which are subjected to high levels of mechanical stress, wear and fatigue during normal activity. These devices include prostheses for hip, knee, ankle, shoulder, spine and elbow joints. Therefore, the introduction of the implant in the body is always associated with the risk of microbial infection. Particularly for the fixation of open-fractured bones and joint revision surgeries.
Infection is a major problem in orthopedics leading to implant failure. Likewise, one of the most significant complications of spinal surgery remains post-operative spinal implant infection (PSII). Depending on microbial virulence, PSII can either manifest early, typically within four weeks of surgery, or delayed, after more than four weeks up to years after surgery. It is a challenging task to treat orthopedic implant infections that may lead to implant replacement and in some cases may result in amputation and mortality. The sources of the infection bacteria include:
- The environment of the operating room
- Surgical equipment
- Clothes are worn by medical staff
- Resident bacteria on the patient’s skin
- Bacteria already residing in the patient’s body
Implant-related infections are the result of bacteria adhesion or subsequent biofilm formation. If bacterial adhesion occurs before tissue regeneration takes place, host defenses often cannot prevent surface colonization for certain bacterial species that are capable of forming a protective biofilm layer. Therefore, inhibiting bacterial adhesion is essential to prevent implant-associated infection, because the biofilm is extremely resistant to both the immune system and antibiotics. For orthopedic implants to yield successful results, the implant material must be habitable by bone-forming cells to favor adhesion of osteoblasts, must be able to hinder the formation of soft connective tissue to inhibit adhesion of fibroblasts, and must be anti-infective to discourage bacterial adhesion. Rapid detection of infection is of paramount importance because delaying the start of treatment of PJI (Prosthetic Joint Infections) may result in the failure of the device.

Infection Management
There are different types of treatments being used currently:
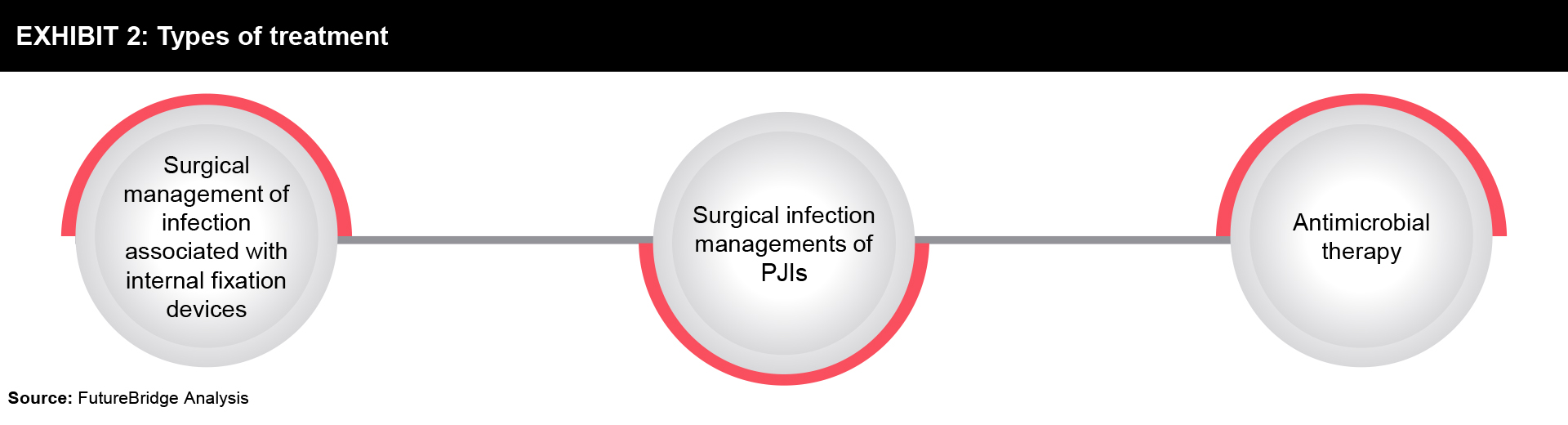

Surgical management of infection associated with internal fixation devices
In contrast to PJI, wherein the goal is to alleviate suffering, restore normal joint function and eliminate the infection, here complete eradication of infection associated with internal fixation devices is not the primary goal until the persistent biofilm does not impair bone healing. Complete healing generally occurs as soon as the foreign material is removed after fracture consolidation. The stability of the fracture is crucial for preventing and treating device-associated infection. Therefore, the maintenance of stable internal fixation devices has been suggested; however, success rates vary between 68% and 100%. This wide variation is probably due to the type of antibiotics used, as the choice of appropriate antimicrobial agents is crucial for the treatment of biofilm infection. The principal techniques of surgical management are debridement and stabilization. The technique of debridement also includes diagnostics biopsy for microbiological and histological evaluation.
Surgical infection management of PJIs (Prosthetic Joint Infections)
The traditional PJI treatment for surgery typically involves the two-stage exchange of the device comprising various interventions and the first intervention includes thorough removal of necrotic tissue, bone cement, and all prosthetic material. The patient is treated with antibiotics during the implant-free period before the new artificial joint is implanted. Because this procedure is invasive and generally leads to functional impairment, less-invasive treatment approaches have been increasingly used.
Antimicrobial therapy
 Antimicrobial treatment comprising antibiotics, without surgery is not recommended and should only be performed if the patient refuses surgery or if the surgical process is associated with a high risk for patient life, in case of different implant-associated infections. Also, in contrast to many other infections such as pneumonia or urinary tract infection, PJIs specifically never spontaneously heal, thus long-term antimicrobial therapy is frequently favored here as well. Until recently, it has been generally considered that PJI cannot be cured without the need for the device because implant adherent bacteria persist as a biofilm. Therefore, successful treatment of implant-associated infection should consider this property of the microorganisms. To date, only two classes of drugs have shown the properties that are needed for the efficacious elimination of biofilm bacteria. Rifampicin and other rifamycins act on biofilm staphylococci, and fluoroquinolones on gram-negative bacilli.
Antimicrobial treatment comprising antibiotics, without surgery is not recommended and should only be performed if the patient refuses surgery or if the surgical process is associated with a high risk for patient life, in case of different implant-associated infections. Also, in contrast to many other infections such as pneumonia or urinary tract infection, PJIs specifically never spontaneously heal, thus long-term antimicrobial therapy is frequently favored here as well. Until recently, it has been generally considered that PJI cannot be cured without the need for the device because implant adherent bacteria persist as a biofilm. Therefore, successful treatment of implant-associated infection should consider this property of the microorganisms. To date, only two classes of drugs have shown the properties that are needed for the efficacious elimination of biofilm bacteria. Rifampicin and other rifamycins act on biofilm staphylococci, and fluoroquinolones on gram-negative bacilli.
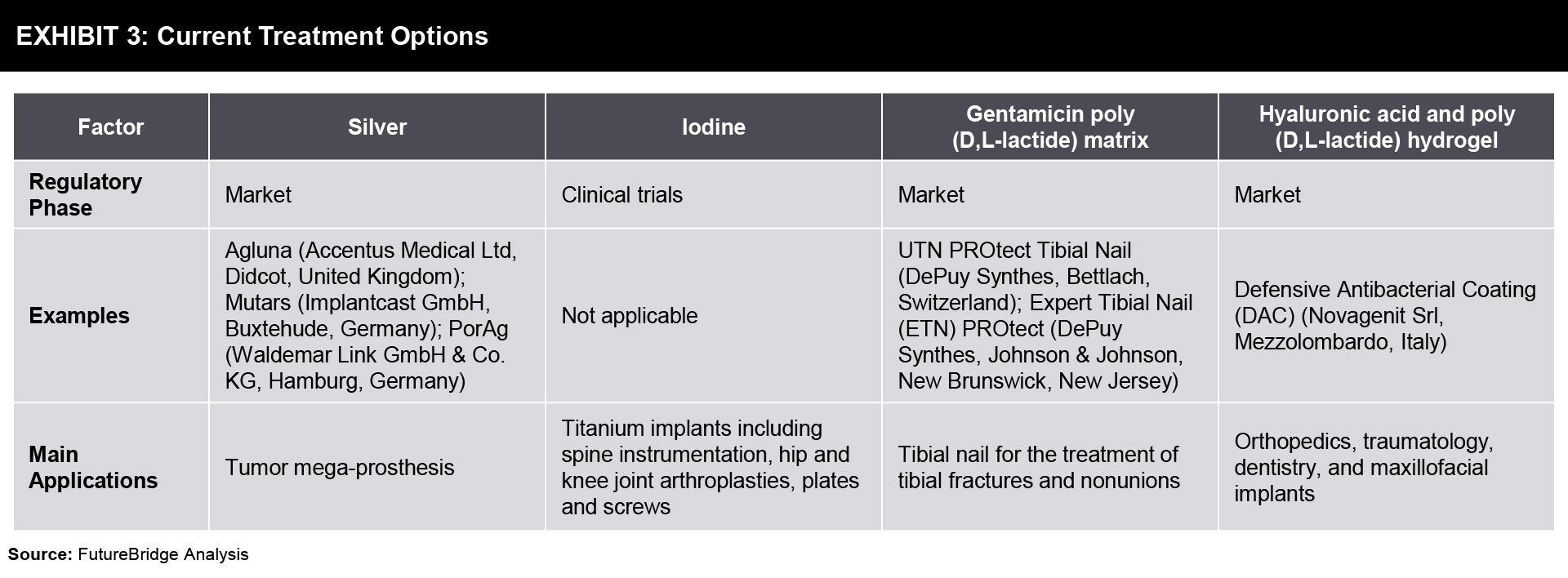
Current treatment concepts are based on the surgical removal of the infected tissue and strict antibiotic treatment to reduce the bacterial burden as much as possible. Antibiotic regimens depend on the result of susceptibility testing of isolated cultures and should be administered for a total duration of 6-12 weeks. One of the most reliable preventive measures available is antimicrobial coating technologies specifically designed for orthopedics and trauma implants. Some companies are making these coatings at the clinical level.

Future Prospects
Novel intervention strategies to either prevent or treat orthopedic implant related infections are the future.
Preventive Approach– Various technologies to be adopted to prevent implant infection are still evolving. But some companies are actively working to develop such futuristic technologies e.g., Pravibismane, a lead therapeutic candidate of Microbion Corporation, is a new class of anti-infective drugs having the ability to prevent and eradicate biofilms that contribute to chronic infection and facilitate antibiotic-resistance. Some examples include:
- Smart coating that gets degraded when bacteria are present and uses nano/micro sensors to detect bacteria.
- Combination Therapy
- Modified Surfaces like super hydrophobic/hydrophobic coatings, black silicon (e.g., Dragonfly wing), and Silicon nitride
- New Antimicrobials like Bismuth thiols (e.g. Microbion); Seldox (e.g Selenium Limited/Emergent technologies); cis-2-deceonic acid (e.g. Disperse biofilms by Dr. David Davis); Imidazoles (e.g. Disperse biofilms by Agile Sciences); Dispersin B (e.g. Inhibit and disperse biofilms by Kane biotech); Quorum sensing inhibitors (e.g. Inhibit biofilms by Dr. Bonnie Bassler); CZ compounds (e.g. Inhibit, disperse and kill biofilms by Curza)
- Dispersing MRSA (Methicillin-Resistant Staphylococcus Aureus) biofilms
Treatment Approach– Current curative approaches like radical debridement, revision surgery, and prolonged antibiotic therapy often result in socio-economic costs, not to mention the risk of life-long functional impairment for the patient. Against this background, orthopedic implant-related infection is set to pose a challenge for practicing clinicians. Various therapies and possible solutions to look out for in the future are:
- Systemic/Local Antibiotic therapy
- Active and Passive vaccines – Multiple attempts are on to create vaccines for S. aureus infection, wherein to date the passive S. Aureus vaccines such as Altastaph, Veronate, Aurexis, Aurograb, and Pagibaximab have failed in clinical trials. In the future, the development of such vaccines holds potential.
- Antimicrobial and anti-biofilm peptides- Immobilization of antimicrobial peptides (AMP) on surfaces has been performed with a variety of peptides, and many different chemistries.
- Quorum-sensing inhibitors and biofilm degrading enzymes
- Use of Silver for coating devices, as it provides a broad spectrum of antibacterial activity. Though the controlled release of silver is a challenge with knowledge and optimization of formulations, silver seems a promising addition to our antibacterial arsenal in the fight against device infection.

Conclusion
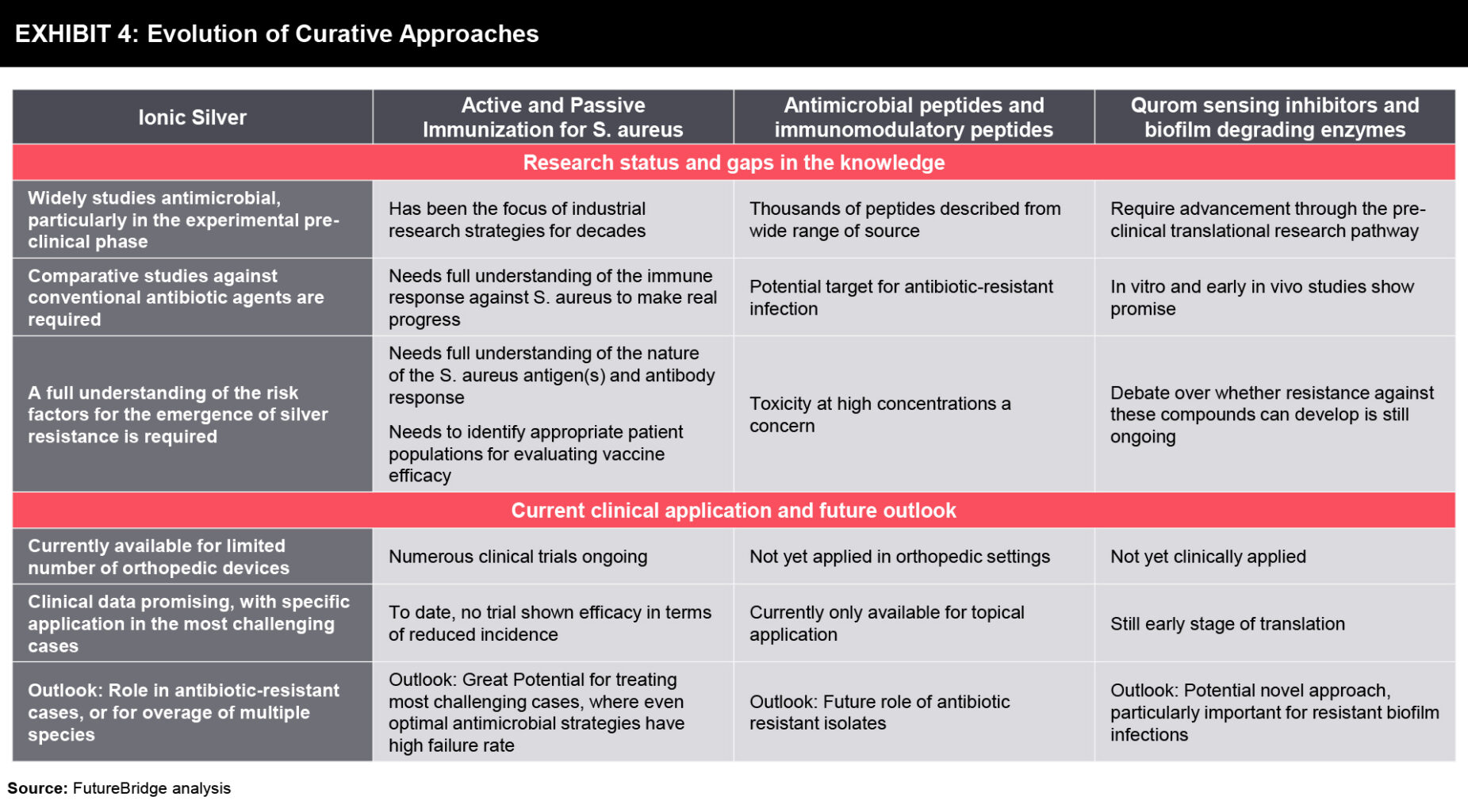
Implant-related infections are a major problem in orthopedic and trauma surgery, and it is expected that the incidence of prosthetic joint infections will further increase in the near future as the number of joint replacements is rising, and complications such as bone infections are also increasing. Certain metallic implants also produce side effects such as physical, electro-chemical irritations, oligo dynamic/catalytic, and carcinogenic effects, which are other potent causes of bacterial infections. Current treatment options are associated with severe consequences for patients and often fail to eliminate the infection. The high risk of chronicity for such infections is due to successful evasion strategies of bacteria with biofilm formation being one major mechanism behind bacterial persistence. Infections that are associated with internal fixation and with prosthetic joints have similar clinical properties. The risk of an implant-associated bacterial infection is especially high with S.aureus bacteremia. Infections associated with internal fixation devices can be treated with implant retention as long as the fracture is stable. By contrast, retention of an artificial joint is only successful in the case of acute haematogenous or early postoperative PJIs. Rifampicin is still the only drug with high activity against gram-negative bacilli associated with orthopedic implants. Surgical treatment should follow a well-defined treatment algorithm. Orthopedic implant infection management includes strategies such as ionic silver, active and passive immunization for S. aureus, antimicrobial peptides, immunomodulatory peptides, quorum sensing inhibitors, and biofilm degrading enzymes. Prevention may be achieved in the future by coating implants with novel antimicrobials. Novel antimicrobial agents that act specifically on established implant adherent biofilms are the need of the hour. Rationally designed, custom 3D-printed antibiotic-impregnated spacer technologies have emerged that can achieve both initial high-dose bolus release and sustained antibiotic release at levels above the minimum inhibitory concentrations (MIC).
References
- Chronic implant related bone infections
- Graphite nanoplates prevent infections
- Clinical presentation and treatment of orthopaedic implant associated infection
- Diagnosis and management of orthopaedic implant associated infection
- Infection of orthopaedic implants with emphasis on bacterial adhesion process and techniques used in studying bacterial-material infections
- Surface treatment strategies to combat implant related infection from the beginning
- Medical device technologies: Potential to treat and prevent biofilm implant related infections
- Microbion
- Orthopaedic device related infection: Current and future interventions for improved prevention and treatment
- Clinical presentation and diagnosis of delayed postoperative spinal implant infection
- Evolving concepts in bone infection
- Management of periprosthetic joint infection
Need a thought partner?
Share your focus area or question to engage with our Analysts through the Business Objectives service.
Submit My Business ObjectiveOur Clients
Our long-standing clients include some of the worlds leading brands and forward-thinking corporations.
- © 2021 Cheers Interactive (India) Private Limited. All rights reserved. FutureBridge ® is a registered trademark of Cheers Interactive (India) Private Limited.




































